4 Polymers
Chapter Outline
- 3.1 Introduction
- 3.2 Learning Objectives
- 3.3 Fundamentals of Polymers
- 3.3.1 Monomers
- 3.3.2 Polymer Structure
- 3.3.3 Additives
- 3.4 Thermoplastics and Thermosetting Polymers
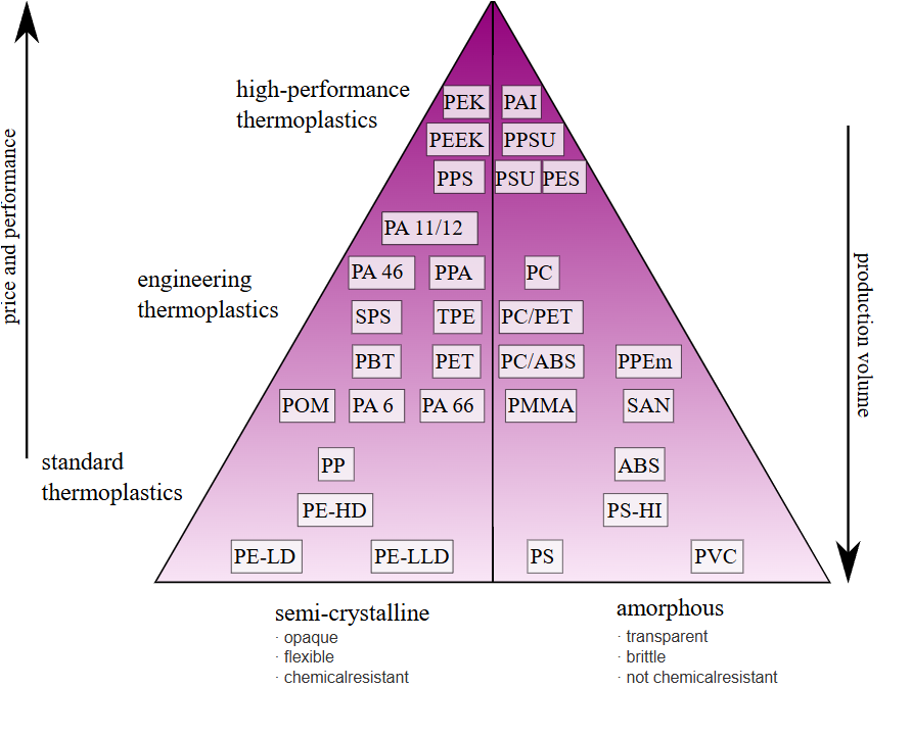
- 3.4.1 Properties of Thermoplastics and Thermosets
- 3.4.2 Common Thermoplastics
- 3.4.3 Common Thermosets
- 3.5 Elastomers
- 3.5.1 Natural Rubber
- 3.5.2 Other Common Elastomers
- 3.6 Polymer Recycling and Degradation
- 3.6.1 Recycling: thermoplastics and more
- 3.6.2 Biodegradable polymers
- 3.7 Chapter Summary
- 3.8 References
3.1 Introduction
The word polymer can refer both to the large macromolecule composed of many repeating subunits called monomers, as well as the substance made up of these macromolecules. This broad definition leads the category of polymers to be vast, containing many essential materials common to use in engineering, manufacturing and everyday life. Organic polymers, such as proteins (polyamides) or carbohydrates (polysaccharides) have essential roles in biology, which are beyond the scope of this chapter. We focus instead on common polymers used in engineering and manufacturing, which are primarily synthetic polymers. There are 3 common types of polymers we will cover: Thermoplastics, thermosets and elastomers.
3.2 Learning Objectives
After reading this chapter, you will be able to:
- Understand the basic fundamentals of polymer chemistry
- Identify polymer structure and its effects on the polymer macromolecule
- Understand the three basic categories of polymers: thermoplastics, thermosets and elastomers
- Identify common polymers used in manufacturing
- Describe polymer recycling and biodegradation
3.3 Fundamentals of Polymers
3.3.1 Monomers
As noted in the introduction, monomers are the repeating units which compose polymers. By definition, monomers must be unsaturated and able to polymerize. For our purposes, that means being able to bond to 2 other monomers to form a chain. These primary polymer bonds are covalent bonds. A monomer that can only form these 2 connections is called a bifunctional monomer. However, many monomers can form additional connections that link sections of the monomer chain to itself or to other chains, forming networks in the resulting polymer.
Polymers are often named after the monomers that comprise them, for example polyvinyl chloride (PVC) is made up of chains of vinyl chloride molecules. Being made up of only one monomer, PVC is known as a homopolymer. A polymer composed of 2 or more different monomers is known as a copolymer. Specifically, polymers composed of 2 monomers are called biopolymers, ones composed of 3 monomers are called terpolymers, and those made of 4 monomers are referred to as quaterpolymers.
3.3.2 Polymer Structure
The primary structure of a polymer is a chain of covalently bonded monomers. The monomers that compose the polymer contain functional groups which change the structure of the polymer, imparting emergent properties. In this section we explore major topics in polymer structure.
Length:
Length, or degree of polymerization, refers to the number of monomers in a polymer chain. Commonly abbreviated as n, for the number of monomers, this value has many effects on the resulting polymer. In a polymeric macromolecule, the degree of polymerization will differ from molecule to molecule, so n is the mean number of monomers and typically ranges from hundreds to thousands. While it makes intuitive sense, it is worth noting that as chain length for a polymer of identical composition increases, the molecular weight of the polymer increases. Additionally, as n increases, the mechanical strength and melting point of the polymer increases.
Configuration:
Configuration refers to the arrangement of monomers in the polymer chain. A homopolymer is composed of only one monomer. Thus homopolymers have only one configuration. Configuration becomes more complicated for copolymers. Copolymers may have an alternating distribution, where one, then the other comonomer are alternated in the polymer chain. There is also random distribution, where the copolymers are distributed randomly throughout the polymer chain. Another distribution pattern is called block, due to “blocks”- chains of one copolymer inserted into another copolymer. Graft configuration occurs when a chain of one copolymer is grafted at a joint onto a chain of another copolymer. In gradient distribution, the chain progressively contains more of one monomer than the other. Table 3.1, below, shows these configurations with different monomers represented by the symbols A and B.
| Polymer Type | Pictorial Representation |
| Random copolymer | |
| Alternating copolymer | |
| Gradient copolymer | |
| Block copolymer | |
| Graft copolymer | 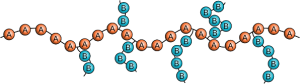 |
Cross Linking:
As previously mentioned, the primary bonding of polymer chains is covalent bonding. In some polymers (thermosets and elastomers), the monomers form additional covalent bonds with adjacent polymer chains, interlinking the polymer with strong bonds that create a structured network. Since this is the same primary bonding that occurs between monomers, a polymer with high cross linking is one large molecule.

Tacticity:
Tacticity, or stereoregularity, is a measure of the regularity of the orientation of monomer side groups on the “backbone” of the polymer chain. Isotactic macromolecules are all oriented in the same direction. Monomers in syndiotactic polymers have alternating orientations. Finally, atactic monomers are oriented in random directions. Tacticity changes the way polymer chains interact with itself and other polymers, and thus has a large role in crystallization, which we will discuss next.
Crystallization:
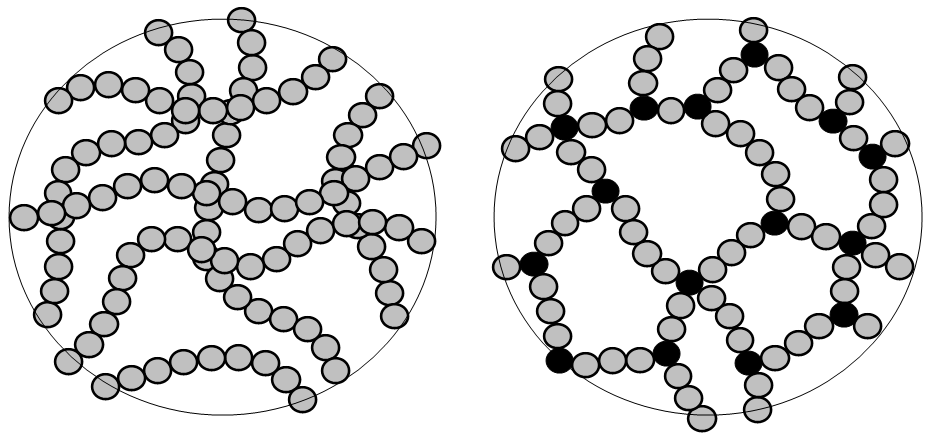
The natural state of many polymers is an amorphous solid, but polymers can form areas of crystal structure through parallel alignment of polymer chains. The length of polymer chains and number of chains present make it impossible for the entire polymer to achieve 100% orderly crystal structure. For common polymers relevant to engineering, crystallinity, which is measured as the ratio of crystallized to uncrystallized polymers by weight or mass, generally ranges from 0-0.8. Thus crystalline polymers are more accurately called semi crystalline. Increased crystallinity in polymers is generally associated with increased material strength, as well as opacity- polymers with higher crystal content tend to be opaque, while amorphous polymers can be transparent.
 |
 |
 |
| Structure of an amorphous polymer | Structure of a semi-crystalline polymer | Structure of a polymer with both amorphous and crystalline regions |
Crystal formation depends on several factors related to polymer structure. Isotactic polymers have a more regular structure that lends itself to forming the parallel chain structure of polymer crystals. Conversely it is more difficult to crystalize atactic polymers, particularly those with large side groups on their monomers, such as rubber. Cross linking holds polymer chains in place in a covalent bonded network, which doesn’t allow the polymer chains to slide past one another and align into crystals. This means thermosets and elastomers cannot crystalize.
The primary method of crystallization in polymers occurs as thermoplastics cool after being heated during processing. Crystals begin with nucleation, as the chains align due to random motions of the heated polymer. This nucleation site can either be destroyed or compounded by the addition of further parallel polymer chains.
3.3.3 Additives
Copolymers:
As briefly discussed previously, copolymers are polymers composed of multiple types of monomers. Many common polymers are copolymers, such as ABS, nitrile rubber and styrene.
The addition of different monomers allows the manufacturer to change the properties of the polymer macromolecule and thus the final product. Common copolymers improve the mechanical strength of finished products. They can also reduce crystallization, or improve the thermal or chemical resistance of the copolymer they form.
Plastisizers:
One of the properties polymers are prized for in manufacturing is its plasticity- its softness and flexibility. This allows the final product to flex without breaking, an important characteristic for polymers used in making pipes, building materials and wire insulation. Additionally, less brittle materials are easier to manufacture and transport without damage. Plasticizers, the most common polymer additive, can be added to improve this property.
Fillers:
Fillers are additives that are added primarily to lower the cost of the polymer. Common fillers are calcium carbonate, carbon, glass fibers, starch, cellulose fibers and silica. While their primary purpose is to reduce costs, fillers are often chosen to complement the polymers properties. They may add strength, rigidity or improve the resistance of the polymer to UV or temperature changes.
Other Additives:
There are many other additives that are commonly added to polymers.
- Colorants are added to many polymers to change their appearance.
- Being derived from petroleum, a lot of polymers are flammable. Flame retardant additives can help prevent or slow the polymer burning.
- Additives can also help polymers resist high temperatures, a typical weakness of polymers. These additives are called heat stabilizers.
- In addition to heat, many polymers are weak to UV light. Light stabilizers can protect polymers from this kind of damage. These additives can come in the form of a coating which does not allow UV to penetrate the part, molecules that disrupt the energy of UV light, or additives that prevent the polymer from being degraded by holding the polymer chains together.
3.4 Thermoplastic and Thermosetting Polymers
3.4.1 Properties of Thermoplastics and Thermosets
The three large divisions of polymers are thermoplastic, thermosets and elastomers. This section focuses on thermoplastics and thermosets, two categories of polymers defined by their reaction to heating.
Thermoplastics:
Thermoplastics, or thermosoftening plastics, are polymers that soften and are able to be formed or reformed after heating. This property is the result of the structure of the polymer. Thermoplastics have polymer chains which interact with each other by weak intermolecular forces (Van der Waals), which are significantly weaker than the primary covalent bonds of the polymer chain. As a thermoplastic, which generally have lower melting points than thermosets, is heated, these forces become weaker and the polymer chains can slip past one another. This allows the polymer to act as a liquid, capable of common manufacturing techniques such as injection molding, compression molding, calendaring, and extrusion.
Thermoplastics do not form strong covalent bonds between polymer chains, which makes them generally weaker and less resistant to heating, stress and strain. Additionally this gives thermoplastics a higher modulus of elasticity, and greater ductility than thermosets. This lack of networked connection allows thermoplastics to be heated and reformed with minimal degradation (discussed further in 3.6 Polymer Recycling and Biodegradation). However, the lack of rigid structure the network provides allows thermoplastics to align into crystalline structures, which thermosets are not capable of.
Thermosets:
Thermosets are differentiated from thermoplastics by their networked covalent structure. Strong cross linking between polymer chains fuse the molecules that make up the macromolecule, giving strength to the resultant structure. Thermosets are consequently stronger and more rigid than thermoplastics, but they are also more brittle. Importantly, this fusion is permanent, and thermosets cannot be recycled as conveniently as thermoplastics. While thermosets can melt, their melting points are a higher temperature at which the covalent bonds making up the polymer chains themselves are destroyed (More on this in 3.6 Polymer Recycling and Biodegradation).
3.4.2 Common Thermoplastics
Acrylic
Polymethyl methacrylate is an inexpensive, strong, lightweight thermoplastic used in many engineering applications. It is sold commercially as Plexiglas, Perspex, Lucite, and under many other names. Its density is about half that of glass, which it often acts as a shatter-resistant replacement for.

Polyvinyl Chloride (PVC)
PVC is the third most commonly produced synthetic polymer in the world annually. It is commonly used in pipes, construction materials and packaging such as bottles. With the inclusion of plasticizers it can become very flexible, and can be used to create flexible tubes, insulation for wires, or inflatable items.

Polystyrene
Made of styrene monomers, polystyrene comes in solid or foam forms. The solid form is a transparent, brittle plastic. The foamed form, commonly known as styrofoam, is used as packaging material, as in the image below.

Polylactic Acid (PLA)
PLA is a common biopolymer derived from fermented plant starch. It has a low melting point and thermal expansion, leading to its widespread use as a filament for FDM 3d printing. Its biological origin also allows specific forms of PLA to be used in medical implants, as it degrades into biologically safe chemicals.

Polyethylene (PET)
Polyethylene is the most commonly produced plastic. This is due to its low cost and resistance to water and chemicals. It has many forms, with many additives that can change its properties for the application. It is also noted for its electrical insulation, and used in wire insulation as well as capacitors for this purpose.

Acrylonitrile Butadiene Styrene (ABS)
ABS is a terpolymer made of styrene, acrylonitrile and butadiene. The ratio of these monomers changes the properties of the final polymer. For a common polymer, ABS has excellent mechanical properties, and is thus used in many applications that require lightweight durability.

Nylon
Nylon was the first commercially successful synthetic polymer, synthesized by Wallace Hume Carothers while working for Dupont in 1935. It is commonly used in its fibrous form in many clothing products. Its solid form is used for low friction gears.

Polypropylene
The second most commonly produced synthetic polymer, polypropylene has similar properties to polyethylene. With a density around 0.9 g/cm3, polypropylene is the least dense of the common polymers.

3.4.3 Common Thermosets
Polyurethanes
Unlike previously discussed polymers, polyurethanes are not a specific type of polymers; rather the name refers to the class of polymers characterized by the presence of urethane, a part of the polymer chain formed during polymerization. Most polyurethanes are produced as rigid or flexible foams, although it is also used in creating coatings, adhesives, sealants and in other applications.

Polyepoxides (Epoxy resin)
Another class of polymers, epoxies are characterized by their inclusion of epoxide functional groups. They are known for their high degree of cross linking, lending high mechanical strength to the finished polymer. The polymerization reaction for epoxies is highly exothermic and catalyzed by heating, a positive feedback mechanism that requires additives that slow the reaction if the volume to be cured is too large.

Polyester resin
Polyester resins are commonly used as a cheaper alternative to epoxy resins, since they share many physical properties. Polyester resins are weaker and put off more dangerous chemicals when curing than epoxy. Like epoxies, they begin in a liquid form that is cured. This allows them to effectively wet glass fibers before curing. This composite material is inexpensive, strong and resistant to the elements, hence its use in boats and many other construction applications.

Polyimide
While polyimides are a useful category of polymers defined by containing imide groups, their inclusion in this chapter is intended to be representative of high-performance plastics. These polymers have higher resistance to heat, and are generally tougher and more expensive than other polymers.

3.5 Elastomers
3.5.1 Natural Rubber
Polyisoprene, or rubber, is a natural polymer which occurs organically, and is one major exception to our focus on synthetic polymers due to its wide use in industry. Natural rubber has been used historically by humans both in its natural state as well as after being vulcanized. Its primary use is as car tires, as synthetic rubbers which are more inert have replaced it in many applications.
Vulcanization is a process by which elastomers are cured. It is applied to synthetic rubbers as well, but was initially used to refer to the process whereby natural rubber is treated to produce more desirable properties. Natural rubber is very susceptible to changes in temperature, becoming gummy and losing shape when heated, and becoming brittle when cold. Vulcanization improves these properties, making rubber a much more viable material. During curing, natural rubber is heated and a mixture of chemicals called the cure package which contains primarily sulfur is added. The sulfur atoms form cross links between the polymer chains, and other additives catalyze the otherwise slow vulcanization process. Cross linking is associated with greater temperature resistance and mechanical strength.
3.5.2 Other Common Elastomers
Silicone
Based on repeating siloxane units, silicones are commonly manufactured as synthetic rubbers or oils. Silicone products are noted for their thermal stability, resistance to water, oxidation and UV resistance. It is however very permeable to gasses, making it suitable for some applications and ruling out its use in other applications.

Nitrile Rubber
A copolymer of acrylonitrile and butadiene, nitrile retains its plasticity over a high temperature range. This property, along with its resistance to many chemicals including oil and fuel make it suitable for hoses, gaskets and other automotive and aerospace applications. It is also stronger than natural rubber- one reason it is commonly used to create gloves and other PPE.

Polychloroprene
Originally marketed as DuPrene due to its formulation by Dupont scientists, polychloroprene was later renamed neoprene. It is a particularly inert polymer which is produced as a rubber or foamed like in the wetsuit shown in the figure below, or mousepads. Its inertness also lends itself to medical devices where flexibility and comfort are priorities.

Ethylene Propylene Diene Monomer (EPDM)
EPDM is known as a particularly tough, black colored terpolymer. The saturation of the polymer chain gives this polymer increased resistance to heat, water and UV radiation. These properties are why it is commonly used in tough environments to create membranes and seals.

3.6 Polymer Recycling and Biodegradation
3.6.1 Recycling: Thermoplastics and more
One of the major concerns with the widespread use of polymers in manufacturing is the inherent risk the current use of polymers poses to the environment. Many common polymers are derived from petroleum, a fundamentally limited resource. Additionally, while polymers generally are inert and not directly toxic, they can still be harmful to wildlife. Plasticosis is a disease recently discovered in seabirds, characterized by severe scarring in the stomach and caused by the ingestion of plastic.
One solution to this issue is recycling polymers. As previously mentioned, thermoplastics can be remelted. This makes them an ideal candidate for recycling, as they can be collected, shredded and reformed with low temperature heating compared to glass or metal recycling. The diverse chemistry of polymers can make this process difficult. For polymers to be recycled properly, they must be sorted by polymer type. Within each polymer type there is still massive diversity of additives, meaning the resulting recycled polymer has a (likely detrimental) mix of additives from the polymers comprising it. Without careful provenance, recycled polymers are not suitable for applications requiring purity or strength, such as structural parts, food packaging or medical devices. In applications where some strength is required, new polymer can be mixed with recycled polymers to reduce the amount of new polymer required. Another confounding factor is that thermoplastics break down over repeated remelting cycles.
3.6.2 Biodegradable Polymers
Another approach to dealing with the sustainability of polymers is biodegradable polymers. These polymers are either derived from natural products or engineered to be decomposed by microbes. The advantage of biodegradable polymers over recycled polymers is that by producing a new polymer, its chemical composition is known and thus it can safely be used to create food packaging, a common form of disposable plastic.
Biodegradable polymers are designed to be broken down by microorganisms. To facilitate this, they are designed with low degrees of polymerization and non-toxic monomers. Surface coatings that reduce the permeability of polymers are omitted.
3.7 Chapter Summary
- Polymers are chains of monomers
- Polymer structure affects the properties of the polymer macromolecule
- Thermoplastics are polymers that can melt
- Thermosets are polymers that cure and are highly cross linked
- Elastomers can stretch and reform
- Thermoplastics can be remelted and recycled
- Biodegradable polymers are designed to be broken down by microorganisms
3.8 References
Britannica. “Polymer.” Encyclopædia Britannica, 26 Feb. 2024, https://www.britannica.com/science/polymer.
Chem, Roland. “Branched Macromolecule,” Wikimedia Commons, 15 Jan. 2015, https://upload.wikimedia.org/wikipedia/commons/thumb/4/44/Polymerstruktur-verzweigt.svg/362px-Polymerstruktur-verzweigt.svg.png.
“Highly Cross-Linked Polymer (Thermoset),” Wikimedia Commons, 15 Jan. 2015, https://upload.wikimedia.org/wikipedia/commons/thumb/0/02/Polymerstruktur-engmaschig_vernetzt.svg/195px-Polymerstruktur-engmaschig_vernetzt.svg.png.
“Linear, Unbranched Macromolecule,” Wikimedia Commons, 15 Jan. 2015, https://upload.wikimedia.org/wikipedia/commons/thumb/c/c0/Polymerstruktur-linear.svg/347px-Polymerstruktur-linear.svg.png.
“Semi-Crystalline Structure of an Unbranched Polymer,” Wikimedia Commons, 15 Jan. 2015, https://upload.wikimedia.org/wikipedia/commons/thumb/2/27/Polymerstruktur-teilkristallin.svg/225px-Polymerstruktur-teilkristallin.svg.png.
“Slightly Cross-Linked Polymer (Elastomer),” Wikimedia Commons, 15 Jan. 2015, https://upload.wikimedia.org/wikipedia/commons/thumb/b/b7/Polymerstruktur-weitmaschig_vernetzt.svg/195px-Polymerstruktur-weitmaschig_vernetzt.svg.png.
Dodiuk, Hanna, and Sidney H Goodman. Handbook of Thermoset Plastics. Elsevier Inc. Chapters, 28 Nov. 2013.
Groover, Mikell P. Fundamentals of Modern Manufacturing : Materials, Processes, and Systems. Hoboken, Nj, John Wiley & Sons, Inc, 2013.
Jones, Richard, et al. Compendium of Polymer Terminology and Nomenclature IUPAC Recommendations 2008. The Royal Society of Chemistry, 2008.
Macdermott. Selecting Thermoplastics for Engineering Applications, Second Edition,. CRC Press, 26 Aug. 2020.
McNaught, Alan, and Andrew Wilkinson. Compendium of Chemical Terminology. Oxford, Blackwell, 2006.
Olagoke Olabisi, and Kolapo Adewale. Handbook of Thermoplastics, Second Edition. CRC Press, 3 Feb. 2016.
Society Of Automotive Engineers. Composite Materials Handbook. Volume 5, Ceramic Matrix Composites. Warrendale, Pa., Sae International on Behalf of Cmh-17, a Division of Wichita State University, 2017.
“Thermoplastics vs. Thermoset Materials | Plastics Manufacturing.” Advanced Plastiform, Inc., 17 Dec. 2019, https://advancedplastiform.com/thermoplastics-vs-thermoset-materials/.
Wikimedia Commons. “Diving suit neoprene” https://so.wikipedia.org/wiki/File:Diving_suit_neoprene.jpg. Accessed 13 February, 2026.
Greater Midwest Exteriors. “A Comprehensive Look at Top Commercial Roofing Options” https://gm-exteriors.com/blog/what-are-the-best-types-of-commercial-roofing-materials. Accessed 13 February, 2026.
Merten, Christian. “Introduction to Macromolecular Chemistry aka polymer chemistry” Ruhr-Uni Bochem, 2019. https://www.mertenlab.de/downloads/macro/macromolchem_lecture1.pdf. Accessed 19 February, 2026.
Media Attributions
- Polyoxymethylene 3D ball © Jynto is licensed under a CC0 (Creative Commons Zero) license
- Structures_of_macromolecules © Cjp24 is licensed under a CC BY-SA (Attribution ShareAlike) license
- Statistical copolymer 3D © Minihaa is licensed under a CC0 (Creative Commons Zero) license
- Alternating copolymer 3D © Minihaa is licensed under a CC0 (Creative Commons Zero) license
- Gradient copolymer 3D © Minihaa is licensed under a CC0 (Creative Commons Zero) license
- Block copolymer 3D © Minihaa is licensed under a CC0 (Creative Commons Zero) license
- Graft copolymer 3D © Minihaa is licensed under a CC0 (Creative Commons Zero) license
- highly crosslinked polymer © Roland.chem is licensed under a Public Domain license
- amorphous © Christian Merten is licensed under a CC BY-NC (Attribution NonCommercial) license
- Semi-crystalline © Christian Merten is licensed under a CC BY-NC (Attribution NonCommercial) license
- Polymerstruktur-teilkristallin.svg © Roland.chem is licensed under a CC0 (Creative Commons Zero) license
- Acrylic Business Card Holder Four Tier © Retail Display Direct is licensed under a CC BY-NC (Attribution NonCommercial) license
- Fishing cannon 007 © Dewhurst
- Logikus3 © Gmhofmann is licensed under a Public Domain license
- Prusa 3D Printer © marchyman is licensed under a CC BY-NC (Attribution NonCommercial) license
- water_bottles_bottle_plastic_plastic_bottle_plastic_bottles_supermarket-700 © pxhere is licensed under a CC0 (Creative Commons Zero) license
- plastic-case-with-dividers-1024×683 © The Royal Group is licensed under a CC BY-NC (Attribution NonCommercial) license
- Sample of first DuPont nylon, December 1939 – Heritage Exhibit – Longwood Gardens © Daderot is licensed under a CC0 (Creative Commons Zero) license
- Mint box polypropylene lid © en:User:Polyparadig is licensed under a Public Domain license
- Polyurethane3 © Ural-66 is licensed under a CC0 (Creative Commons Zero) license
- Hybridcircuit © Janke is licensed under a Public Domain license
- Chipironier polyester à Ciboure © Jean Larrieu is licensed under a Public Domain license
- Vespel.Werkstueck.P1149524 © Bautsch is licensed under a Public Domain license
- polymer hierarchy © Minihaa is licensed under a Public Domain license
- Silicone-Sealant-1001U_43333-480x360_(4817475284) © Emilian Robert Vico is licensed under a CC BY (Attribution) license
- as.bluenitrilegloves.01.blue_nitrile_gloves.550 © Multi Range is licensed under a CC BY-NC (Attribution NonCommercial) license
- Diving suit neoprene © Mark.murphy is licensed under a Public Domain license
- Single-Ply Membrane Roofing (TPO, PVC, EPDM) © Greater Midwest Exteriors is licensed under a CC BY-NC (Attribution NonCommercial) license
