3 Metals
2.1 Introduction
Metals are an integral part of many different fields of engineering, and manufacturing. Metals are used and designed for many properties. Anything from aerospace grade titanium, known for its high strength to weight ratio. To ITO (Indium tin oxide), this is a conductive compound that is transparent. This has applications in touch screens and solar panels.
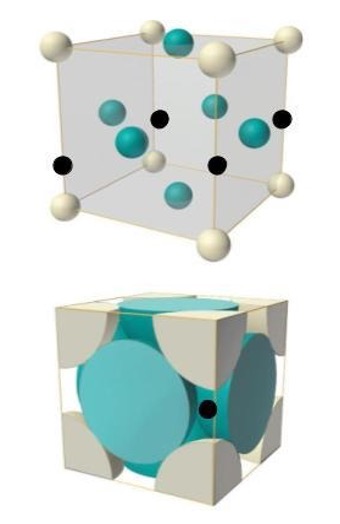
The atomic structure of metals is classified as crystalline structure. This is a repeating lattice of atoms arranged in specific patterns. A unit cell is the smallest repeatable atomic array that is the makeup of solid metals as shown in Fig 1. These unit cells have different structures as shown in both figures. The body-centered cubic unit cell is a more efficient way to pack spheres together and is much more common among pure elements. Each atom has eight nearest neighbors in the unit cell, and 68% of the volume is occupied by the atoms.
These different structures are the building blocks of metal materials.
2.2 Learning objectives
- Understanding different metals structures at different sizes
- Properties of different types of metals
- Phase diagrams used in the industry
- Difference between ferrous and nonferrous metals
- Uses for ferrous metals
- Uses for nonferrous metals
- Understanding what superalloys are and their applications
- Understanding what refractory metals are and their applications
2.3 Metals and Their Phases
2.3.1 Properties of Metals
Steel is an alloy of iron and carbon. This table shows the different carbon percentages (based on the mass of the iron). The more carbon is added to the iron general strength increases, at the cost of the material becoming more brittle.
| Carbon (percent weight compared to Iron) | Alloy |
| 0.25% | Mild Steel |
| 0.25%-0.6% | Medium Carbon Steel |
| 0.6%-2% | High Carbon Steel |
| 2%-4% | Cast Iron |
The next important structure of understanding metals involves the grain. These grains start to form after a metal has heated to a certain temperature. Its crystalline structure starts to reform, they expand in all directions, from different points in the metal, until they collide together. Each of these expanded crystals is referred to as a grain. Many different processes affect grain composition and structure. Heat, and mechanical processes, for example cold pressing can affect these patterns.This is where the material is pressed through rollers at room temperature flattening the material stitching out grains. This increases the ultimate strength of a material but usually lowers ductility. The size and orientation of the grain structure impacts material properties. Generally the smaller a grain structure the stronger the material. This could be achieved through adding precipitants. These allow for more gains to start forming causing them to collide, instead of expanding further before reaching another grain.

2.3.2 Phase Diagrams
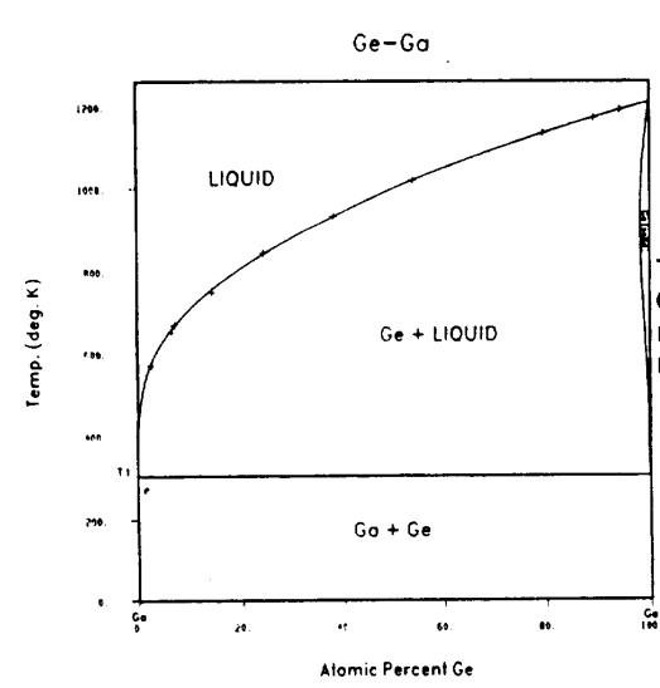
As mentioned metals crystalline structures have different types of unit cells. These cells can not only change with heat but other elements in an alloy. At certain temperatures different metals and alloy structures will change. These relationships of alloys are represented by phase diagrams. On the horizontal axis there is the percent total mass of the added metal. On the vertical axis is the temperature of the material. Different sections of the graph will generally be labeled with the greek alphabet. These usually correspond to the structure of the unit cell (example: FCC) or the phase at the given temperature and material composition.
This specific phase diagram was compiled by Nasa. Their goal was to test the limits of a gallium and germanium alloy. This binary system( two elements) would then be added with arsenic to become a ternary system. This ternary system was used for solar cells in satellites and other electronic space applications. This diagram will show what mission limits this binary system can handle before becoming a liquid, or more importantly finding an ideal mixture of germanium and gallium to handle specific mission requirements.
To explore more about how the unit cell structure and grain structure impacts steel alloys let’s go through a simplified forging process of different heat treatments. For this example a carbon content of .50% was used. Now let’s put this steel into a forge and heat it to approximately 1050 celsius. Now the steel will sit out and slowly cool. After cooling, the form of the steel is a mix of ferrite and cementite. These are both different grain structures on Steel’s phase diagram. Ferrite is a softer iron with a BCC unit cell and cementite is a mix of iron and carbon. Cementite has a greater strength from its carbon content. The carbon atoms cause many dislocations or unevenness in the grain structure. These dislocations allow the material to handle more force. Grain structure will form differently depending on the carbon content of the alloy and temperature.
Now the steel will be heated to the same temperature but rapidly cooled in oil. This traps the steel in a phase called Martensite. This phase has a unit structure (FCC) that allows carbon atoms to fit in between iron atoms in the unit cell. This change of the unit cell in the grain causes the material to become very hard. The downside of this is the ductility is decreased. This is further exaggerated from the fast cooling causing internal tension. The surface was exposed to oil, cooling while still leaving the interior to be higher in temperature. This will cause internal tension in the steel. Now the material can’t release stress by deformation. Once a great enough force is applied the martensite will roughly break in two releasing tension and metal debris.
This FCC diagram shows another example of carbon atoms(black circles) being able to fit in between iron atoms. This also occurs in Austenite, another phase of steel. This allows for higher packing efficiency and denser grain structure.
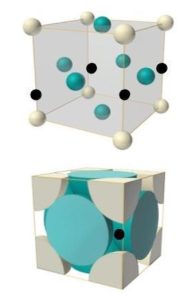
This FCC diagram shows another example of carbon atoms (black circles) being able to fit in between iron atoms. This also occurs in Austenite, another phase of steel. This allows for higher packing efficiency and denser grain structure. Giving the material more strength.
2.4 Difference between Ferrous and Nonferrous Metals
2.4.1 What are They?
Ferrous metals are a category of metals that contain iron as their main constituent element. The most common ferrous metals include carbon steel, alloy steel, stainless steel, cast iron, and wrought iron. These materials are widely used in engineering applications due to their high tensile strength, durability, and resistance to wear and tear. Additionally, ferrous metals are magnetic and conductive, making them useful for a wide range of applications such as construction, automotive, machinery, and aerospace. Understanding the properties and characteristics of ferrous metals is essential for selecting the appropriate material for a given application (Robert Hewitt, “Ferrous Metals”).

Nonferrous metals are metals that do not contain iron as a major component. These metals have different properties and applications compared to ferrous metals. Common nonferrous metals include aluminum, copper, zinc, nickel, and titanium, among others. One of the most notable advantages of nonferrous metals is their corrosion resistance. Unlike iron-based metals, they do not rust, which makes them useful in various applications, especially in corrosive environments. Additionally, nonferrous metals can be lighter in weight, making them ideal for applications that require high strength-to-weight ratios. (Robert Hewitt, “Nonferrous Metals”).
2.4.2 Production and Applications of Ferrous Metals
Steel is a widely used alloy of iron and carbon, with carbon content typically ranging from 0.2% to 2.1% by weight. It is known for its high strength, durability, and versatility, making it a popular choice in a wide range of applications, including construction, manufacturing, and transportation. Steel can be produced in a variety of forms, including flat sheets, bars, tubes, and wire, and it can be easily fabricated and shaped using a variety of techniques, including welding, cutting, and forging. The specific properties of steel can be adjusted through the addition of other elements, such as manganese, nickel, and chromium, allowing for even greater versatility in its use (Robert Hewitt, “Ferrous Metals → Steel”).
Cast iron is a group of iron-carbon alloys with a carbon content greater than 2%. It is known for its excellent castability and unique properties, making it a valuable material in various industrial applications. Cast iron has several advantages, including good castability, wear resistance, and excellent thermal conductivity and vibration dampening. However, it can be brittle and prone to cracking if subjected to sudden impacts or stresses. The choice of cast iron type depends on the specific application and the desired balance of properties, such as strength, ductility, and heat resistance (Robert Hewitt, “Ferrous Metals → Cast Iron”).
Wrought iron is a nearly obsolete type of iron that has been worked by hammering, bending, and shaping while it is in a heated, malleable state. It is characterized by its fibrous appearance, which is a result of the distinctive grain structure formed during the forging process. Wrought iron has been used for centuries in various applications, including decorative ironwork, architectural elements, and tools. Wrought iron has largely declined in modern times due to changes in manufacturing processes. Contemporary materials like mild steel and alloys are often used as substitutes because they are more readily available and easier to produce. However, modern blacksmiths and artisans still work with traditional wrought iron for restoration and preservation of historical structures and for creating authentic, handcrafted pieces (Robert Hewitt, “Ferrous Metals → Wrought Iron”).
2.4.3 Production and Applications of Nonferrous Metals
Aluminum is a lightweight and versatile metal widely used in engineering applications due to its desirable properties. It has excellent strength-to-weight ratio, good corrosion resistance, and can be easily machined, formed, and welded. As a result, it is commonly used in the transportation industry for aircraft, automobiles, and trains. It is also used in packaging, construction, and consumer products such as appliances, electronics, and sporting equipment. Aluminum is available in a variety of alloys, which are designated by a numbering system based on their chemical composition and properties. Each alloy has a unique set of characteristics, making it suitable for different applications (Robert Hewitt, “Nonferrous Metals → Aluminum”).
Copper is a versatile and widely used engineering material due to its excellent electrical and thermal conductivity, corrosion resistance, and malleability. Copper is commonly used in electrical wiring, plumbing systems, heat exchangers, and electronic devices. It is also used in architecture and construction for roofing, gutters, and ornamental features. Copper is available in various forms, including pure copper and copper alloys, such as brass and bronze. Copper alloys provide enhanced properties, including higher strength and improved corrosion resistance. The properties of copper can be further modified through various heat treatment processes. Despite its many advantages, copper has some limitations, such as its susceptibility to oxidation and its relatively high cost compared to other materials (Robert Hewitt, “Nonferrous Metals → Copper”).
Nickel is a versatile and corrosion-resistant metal belonging to the transition metals group. Known for its silvery-white appearance, nickel exhibits excellent properties that make it widely utilized across various industries. In its pure form, nickel is ductile, malleable, and possesses good electrical and thermal conductivity. One notable aspect of nickel is its exceptional resistance to corrosion and oxidation, making it a valuable material for applications in challenging environments. Additionally, nickel is often alloyed with other metals to create alloys with specific properties. One such example is the combination of nickel with chromium to form stainless steel, renowned for its corrosion resistance and durability (Robert Hewitt, “Nonferrous Metals → Nickel”).
2.5 Superalloys
The term ‘superalloy’ came into existence shortly after the second world war and is used to describe a group of materials capable of sustaining their properties even at higher temperatures. Superalloys are uncommon materials because of their excellent physical and chemical properties, such as superior mechanical and thermal strength, good fatigue resistance, better surface stability, and resistance to oxidation or corrosion. Superalloys have gained prominence in recent years in various sectors, namely, spacecraft, marine, power, defense, vehicular and others, due to their ability to withstand high temperatures of up to 980 °C without deformation (Ganesha Prasad).
The commonly used superalloys include Inconel, Haynes, Monel, Nimonic, Incoloy, Waspaloy, Hastelloy, and many more. A superalloy is a combination of metals like Ni, Co, Cr, and Fe from the VIIIB group in the periodic table and some other metals like Al, Ta, Mo, Nb, W, and Ti in lesser proportion. There are three categories of superalloys classified based on more significant percentages of certain metal content, namely, Ni-based, Co-based, and Fe-based. Amongst all the superalloys, Ni-based superalloys have a much better capability to retain the properties at high temperatures near the melting point and have good resistance to oxidation and creep due to the presence of Ni-Al-Ti in the precipitation form in the Ni-Cr matrix (Ganesha Prasad).

2.6 Refractory Metals
Refractory metals are known for their special properties: a high melting point, outstanding mechanical strength, good electrical conductivity, and excellent corrosion resistance. These properties make them the ideal materials for demanding industrial applications in various industries, such as the semiconductor industry, power electronics, e-mobility, and the energy industry (Plansee).
Special Properties of Refractory Metals (Plansee):
- Heat resistance, dimensional stability, and high strength
- Good electrical and thermal conductivity
- Low thermal coefficient of expansion
- High corrosion resistance
- Shielding of X-ray and gamma radiation

2.7 Chapter Summary
Let’s go over some key points, different unit cells and grain structure can be manipulated in different ways to change the material proprietors of steel. These different grain structures are referred to as phases. They can be represented using a phase diagram to show the relationship between temperature and mass percentage of added metal. These grains can have different unit cells which interact with different materials in the alloy.
Ferrous and nonferrous metals both have their own pros and cons. Ferrous metals are metals that have iron as their main element and are widely used in engineering because of their high tensile strength, durability, and resistance to wear and tear. They are also very magnetic and conductive making them very useful in areas such as construction, automotive, machinery and aerospace. Nonferrous metals are metals that don’t have iron as their main element and are known for their corrosion resistance, which makes it very ideal in very corrosive environments. They are also very lightweight compared to ferrous metals.
Superalloys are a group of metals that can sustain their properties at very high temperatures. They have excellent physical and chemical properties, they have superior mechanical and thermal strength, they have a decent fatigue resistance, resistance to oxidation and corrosion, and have a better surface stability. Superalloys are used in many fields, such as spacecraft, marine, power, defense, vehicular and others, due to their ability to withstand high temperatures.
Refractory metals are another group of metals that have high melting points, amazing mechanical strength, good electrical conductivity, and great corrosion resistance. Refractory metals are mainly used in fields such as semiconductor industry, power electronics, e-mobility, and the energy industry.
2.8 References
DuVall, John Barry, and David R. Hillis. Manufacturing Processes: Materials, Productivity, and Lean Strategies. 3rd ed., Goodheart-Willcox, 2011.
The Efficient Engineer. Understanding Metals. 8 June 2021. YouTube, https://www.youtube.com/watch?v=PaGJwOPg2kU. Accessed 23 January 2025.
Groover, Mikell P. Fundamentals of Modern Manufacturing: Materials, Processes, and Systems. 6th ed., Wiley, 2015.
Hewitt, Robert W. “Metals and Alloys.” Engineering Technology, https://engineeringtechnology.org/engineering-materials/metals-and-alloys/ . Accessed 6 February 2025.
Plansee. “Refractory metals are our passion.” Plansee, https://www.plansee.com/en/materials.html?campaignid=1948341588&adgroupid=69113108285&keyword=refractory%20metals&device=c&gclid=EAIaIQobChMI6pKivovBiwMV4iCtBh20wzI6EAAYASAAEgLfpvD_BwE&placement=&gad_source=1. Accessed 13 Febraury 2025.
Prasad, Ganesha, et al. “A review on conventional and nonconventional machining of Nickel-based Nimonic superalloy.” EDPSciences, 21 April 2023, https://mfr.edp-open.org/articles/mfreview/full_html/2023/01/mfreview220071/mfreview220071.html. Accessed 13 February 2025.
“12.2: The Arrangement of Atoms in Crystalline Solids.” Libretexts, 24 Nov. 2013, chem.libretexts.org/Bookshelves/General_Chemistry/Book%3A_General_Chemistry%3A_Principles_Patterns_and_Applications_(Averill)/12%3A_Solids/12.02%3A_The_Arrangement_of_Atoms_in_Crystalline_Solids.
D, Scofield J, et al. “Calculation of Gallium-Metal-Arsenic Phase Diagrams.” NASA. Lewis Research Center, Space Photovoltaic Research and Technology Conference, Aug. 1991, https://ntrs.nasa.gov/citations/19910020900, https://doi.org/10020900/downloads/19910020900. Accessed 9 Feb. 2025.
“Heat Treatment -the Science of Forging (Feat. Alec Steele).” YouTube, 29 Jan. 2018, https://youtube.com/watch?v=6jQ4y0LK1kY.
Wu, Billy. “How to Make Metal Stronger by Heat Treating, Alloying and Strain Hardening.” YouTube, 1 Oct. 2020, https://youtube.com/watch?v=7lM-Y4XndsE. Accessed 1 Oct. 2020.
Media Attributions
- FCC Crystal Structure © Anonymous LibreTexts is licensed under a CC BY-NC-SA (Attribution NonCommercial ShareAlike) license
- Microestuctura Laton © Remux is licensed under a CC BY (Attribution) license
- Ge-Ga Phase Diagram © Scofield, J. D. is licensed under a Public Domain license
- forms of metal © The Closure Library Authors is licensed under a CC BY (Attribution) license
- tensile test bar © Ateekh Ur Rehman, Yusuf Usmani, Ali M. Al-Samhan, Saqib Anwar. is licensed under a CC BY-SA (Attribution ShareAlike) license
- refractory metal © Lander777 is licensed under a Public Domain license
